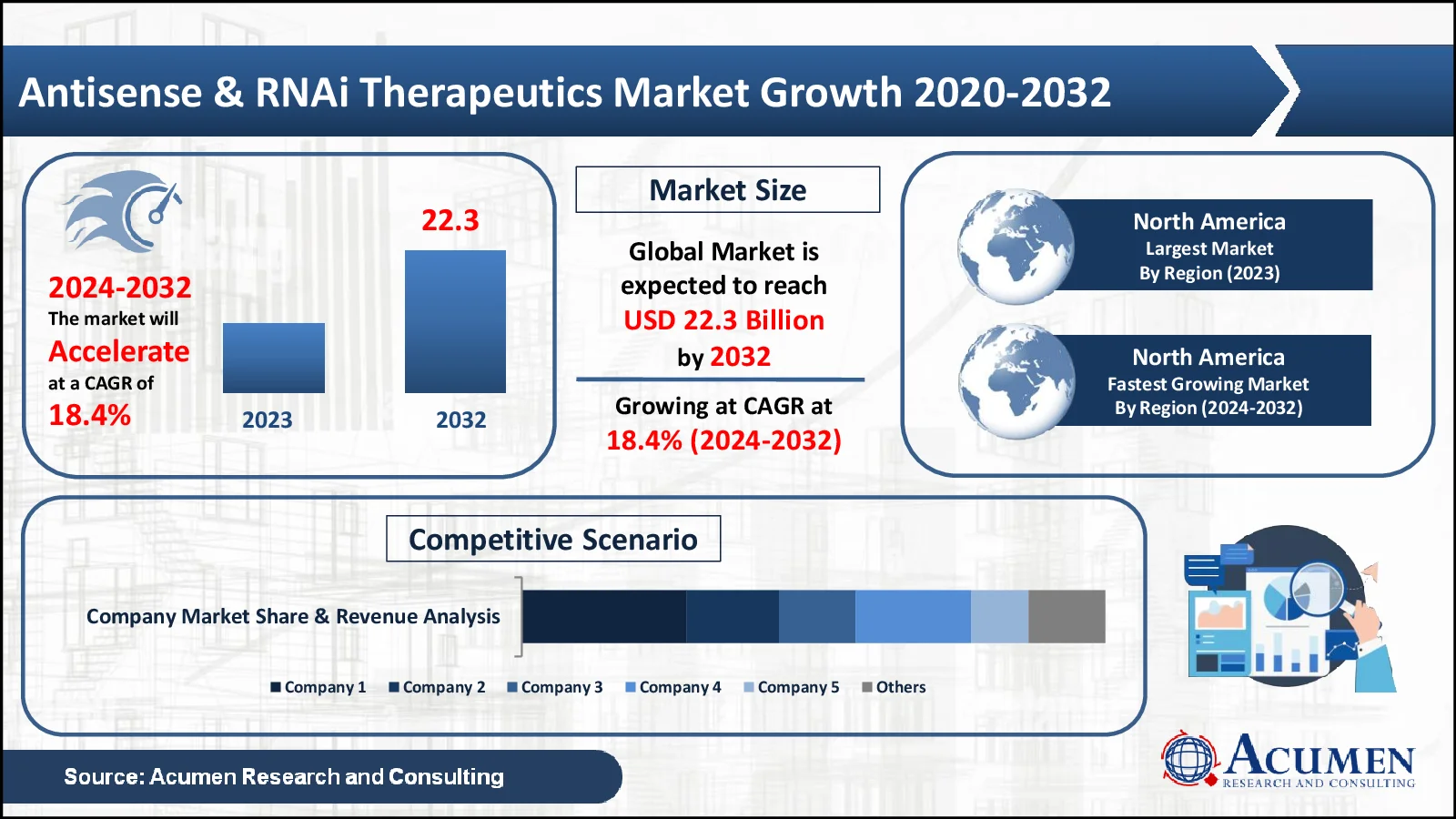
Antisense & RNAi Therapeutics Market Size to Reach USD 22.1 by 2032
The Antisense & RNAi Therapeutics Market, valued at USD 4.9 in 2023, is anticipated to surpass USD 22.1 by 2032, reflecting a projected CAGR of 18.4%
The antisense and RNAi therapeutics market is experiencing robust growth driven by the increasing prevalence of genetic and complex diseases. These therapeutic modalities offer a novel approach to target disease at the genetic level, providing potential for highly specific and effective treatments. Advancements in our understanding of RNA biology and the development of sophisticated delivery systems have significantly propelled this market forward. A key factor driving market expansion is the unmet medical needs in areas such as rare genetic disorders, neurodegenerative diseases, and cancer. Traditional therapeutic approaches often fall short in addressing these complex conditions, making antisense and RNAi therapies particularly attractive. Moreover, the growing body of clinical evidence demonstrating the efficacy of this therapeutics in treating various diseases has fostered increased investor interest and accelerated drug development pipelines.
However, challenges persist in the development of antisense and RNAi therapeutics. Delivery and distribution remain critical hurdles, as these molecules often require specialized delivery systems to reach their target tissues. Additionally, the high cost of drug development and the complexities of clinical trials can pose significant barriers to market entry. Nonetheless, the potential benefits of these therapies and the ongoing research efforts to overcome these challenges are expected to sustain market growth in the coming years. In conclusion, the antisense and RNAi therapeutics market is a dynamic and promising landscape. As our knowledge of RNA biology continues to expand and technological advancements enable more efficient drug development, we can anticipate the emergence of innovative treatments for a wide range of diseases.
Antisense & RNAi Therapeutics Market Statistics
- The global antisense & RNAi therapeutics market is expected to reach USD 4.9 by 2023
- The market is expected to grow at a 18.4% CAGR between 2024 and 2032
- North America accounts for 64% of the market share
- North America is the fastest-growing region, with a growth rate of 19.3%, providing opportunities for industry participants
- The intrathecal injections route of administration makes a substantial contribution to the market
- Neurodegenerative disorders application play an important role in revenue generation
- The market is expanding due to expansion of RNAi and antisense therapeutics into rare and orphan diseases
Access Table Of Content: https://www.acumenresearchandconsulting.com/antisense-and-rnai-therapeutics-market
Antisense & RNAi Therapeutics Market Dynamics
Increasing Prevalence of Genetic Disorders Fuels the Antisense & RNAi Therapeutics Market
The burgeoning prevalence of genetic disorders is a primary catalyst propelling the growth of the antisense and RNAi therapeutics market. These disorders, often characterized by their complex and multifaceted nature, have historically posed significant challenges for traditional therapeutic interventions. The advent of antisense and RNAi technologies has offered a beacon of hope in addressing this unmet medical need.
By targeting specific disease-causing genes at the RNA level, these therapeutic modalities have the potential to disrupt the underlying pathophysiology of genetic disorders with unprecedented precision. This targeted approach is particularly advantageous in conditions where the root cause is a genetic mutation, as it offers the prospect of disease modification rather than mere symptom management. As our understanding of the genetic basis of various diseases deepens, the demand for effective and targeted therapies continues to soar.
Moreover, the growing awareness of genetic disorders among the general public and healthcare providers has led to increased early diagnosis and patient advocacy. This heightened focus on genetic diseases has spurred research and development efforts in the antisense and RNAi space, resulting in a robust pipeline of potential treatments. The ability of these therapies to address a wide spectrum of genetic conditions, from rare monogenic disorders to complex polygenic diseases, further underscores their potential to revolutionize patient care.
Expansion into Novel Therapeutic Areas Offers Significant Antisense & RNAi Therapeutics Market Opportunities
The antisense and RNAi therapeutics market presents a vast opportunity for expansion into novel therapeutic areas beyond the traditional focus on rare genetic disorders. The versatility of these technologies, coupled with the growing understanding of RNA biology, has opened doors to explore their potential in addressing a diverse range of diseases.
One promising avenue lies in the field of oncology. Cancer is a complex disease characterized by genetic aberrations, making it an ideal target for antisense and RNAi-based therapies. These modalities offer the potential to inhibit cancer cell proliferation, induce apoptosis, and modulate the tumor microenvironment. Additionally, the ability to target multiple oncogenes simultaneously holds the promise of overcoming drug resistance, a significant challenge in cancer treatment.
Furthermore, the application of antisense and RNAi technologies to inflammatory diseases is gaining traction. These conditions are often driven by dysregulated gene expression, and these therapies can be employed to silence pro-inflammatory genes or enhance the expression of anti-inflammatory factors. By targeting specific RNA molecules involved in the inflammatory cascade, it is possible to develop novel treatments with improved efficacy and safety profiles. As research progresses and the clinical validation of these approaches strengthens, the antisense and RNAi therapeutics market is poised to capitalize on the substantial opportunity presented by these and other emerging therapeutic areas.
Antisense & RNAi Therapeutics Market Segmentation
The global antisense & RNAi therapeutics market is splits into 4 categories: technology, route of administration, application, and regional markets
- Technology: antisense RNA, and RNA interference
- Route of Administration: intrathecal injections, subcutaneous injections, intravenous injections, and other delivery methods
- Application: oncology, cardiovascular, respiratory disorders, renal diseases, neurodegenerative disorders, genetic disorders, infectious diseases, and other
- Regional industry: Latin America, North America, Europe, Asia-Pacific, and the Middle East & Africa
Antisense & RNAi Therapeutics Market Regional Outlook
The antisense and RNAi therapeutics market exhibits distinct regional characteristics, influenced by factors such as healthcare infrastructure, regulatory landscape, research and development investments, and disease prevalence. North America, comprising the United States and Canada, has historically been at the forefront of the antisense and RNAi therapeutics market. The region boasts a robust biotechnology and pharmaceutical industry, coupled with substantial investments in research and development. A favorable regulatory environment and early adoption of innovative therapies have contributed to the dominance of North America.
Antisense & RNAi Therapeutics Market Players
Antisense & RNAi therapeutics companies profiled in the report include GlaxoSmithKline plc, Novartis AG, Arrowhead Pharmaceuticals, Inc., Ionis Pharmaceuticals, Inc., CRISPR Therapeutics, Benitec Biopharma Inc., BioNTech SE, OliX Pharmaceuticals, Inc., Arbutus Biopharma, and Alnylam Pharmaceuticals, Inc.
Enquire Before Buying https://www.acumenresearchandconsulting.com/inquiry-before-buying/1062
Receive our personalized services and customization by clicking here https://www.acumenresearchandconsulting.com/request-customization/1062
Mr. Richard Johnson
Acumen Research and Consulting
India: +91 8983225533
E-mail: [email protected]


