T-Cell Therapy Market Size to Reach USD 23.3 Billion by 2032 growing at 23.8% CAGR - Exclusive Report by Acumen Research and Consulting
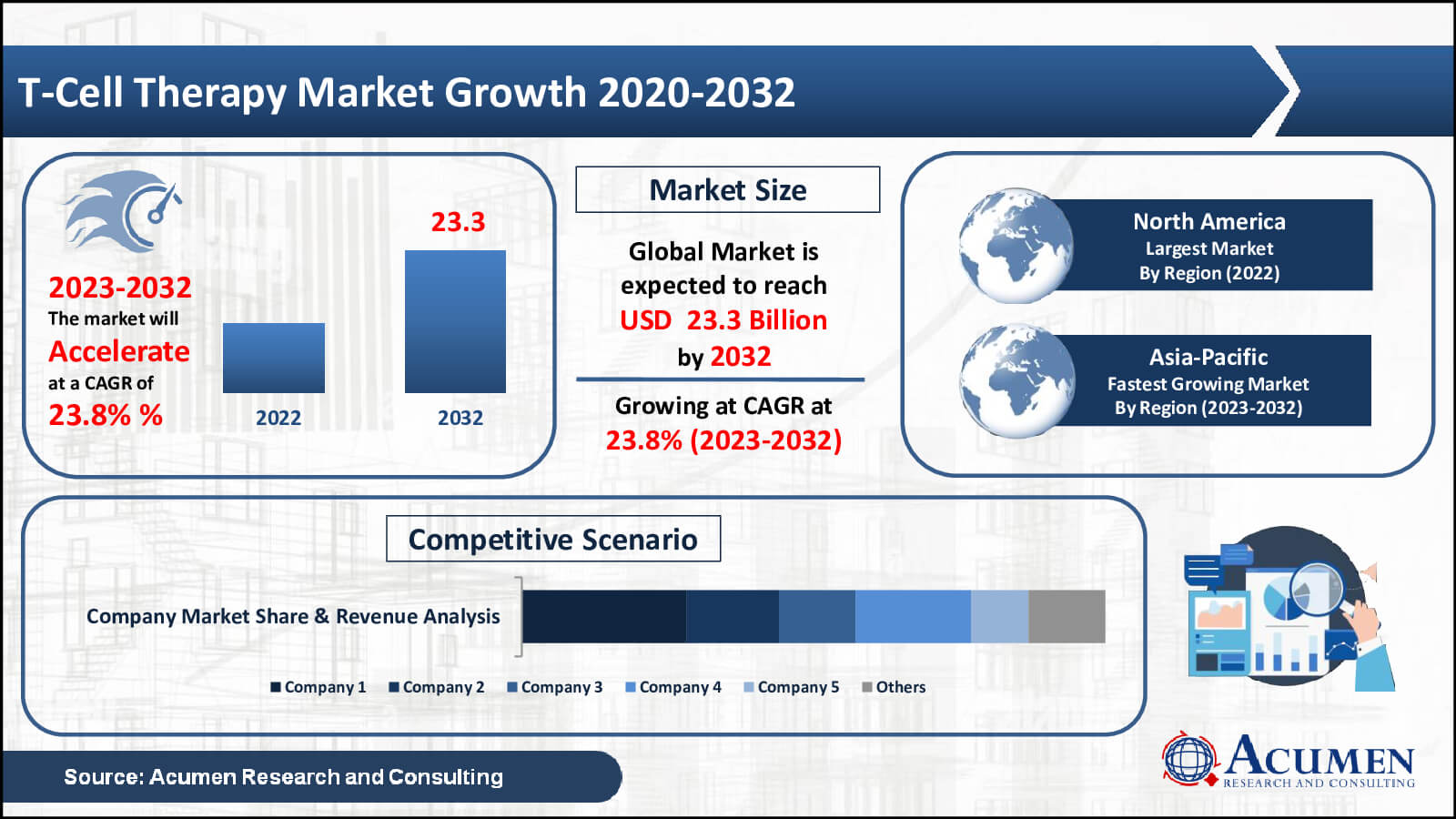
According to Acumen Research & Consulting, the global T-Cell Therapy Market achieved USD 2.8 Billion in 2022 and is projected to surge to around USD 23.3 Billion by 2032, with a substantial CAGR of 23.8% during the forecast period
The T-cell therapy industry has been rapidly evolving as a groundbreaking approach to treating various diseases, particularly cancer. Chimeric Antigen Receptor T-cell (CAR-T) therapy has emerged as a frontrunner, with FDA approvals and successful clinical outcomes in certain blood cancers. The sector's growth has been driven by a surge in investment from major pharmaceutical companies, fostering intense competition and innovation. However, the industry faces challenges, including high treatment costs, safety concerns, and the need for comprehensive regulatory frameworks to ensure patient safety. Beyond oncology, T-cell therapy is diversifying its applications, showing promise in treating autoimmune disorders and infectious diseases. As research and development efforts continue to expand globally, the T-cell therapy industry is poised for continued growth and evolution, with the potential to transform the landscape of healthcare treatments. For the latest developments in this rapidly changing field, it is advisable to consult more recent sources.
T-Cell Therapy Market Statistics
- The global T-cell therapy market is set to reach USD 2.8 billion by 2022, with a steady 23.8% expected CAGR from 2023 to 2032
- North America led in 2022 with over 63% market share
- Asia-Pacific is poised for robust growth, expecting a CAGR of over 25% from 2023 to 2032
- CAR t-cell therapy dominated in 2022, making up over 94% of the market
- Based on indication, hospitals occupied US $ 1.4 billion in 2022
- Facility expansion for cell and gene therapies is a key trend in the T-cell therapy market
Request for a sample of this premium research report@ https://www.acumenresearchandconsulting.com/request-sample/2494
T-Cell Therapy Market Dynamics
Increased Investment In Adoptive T Cell Transfer Approaches To Disease Treatment is Fueling the Worldwide T-Cell Therapy Market Value
Increased investment in adoptive T cell transfer approaches to disease treatment signifies a growing recognition of the potential of T cell-based therapies in revolutionizing healthcare. This approach involves harnessing a patient's own T cells, modifying them, and then reintroducing them into the patient's body to target and combat diseases, particularly cancer. One key driver behind this investment surge is the remarkable success of Chimeric Antigen Receptor T-cell (CAR-T) therapies in treating certain types of leukemia and lymphoma. These therapies have shown impressive response rates and even long-lasting remissions in some patients, prompting considerable interest from pharmaceutical companies and investors.
Furthermore, the broader adoption of adoptive T cell transfer approaches extends beyond CAR-T therapy. Researchers are exploring the use of T cell receptor (TCR) therapies and tumor-infiltrating lymphocytes (TILs) for a wider range of diseases, including solid tumors. This diversification of applications broadens the potential market and therapeutic possibilities, attracting increased investment.
However, it's essential to acknowledge the challenges associated with adoptive T cell transfer therapies. High production costs, complex logistics, and safety concerns, such as cytokine release syndrome and neurotoxicity, have been notable hurdles. Therefore, part of the investment is directed towards addressing these challenges, with ongoing research efforts focused on enhancing the safety and scalability of T cell therapies. In summary, the increased investment in adoptive T cell transfer approaches reflects the industry's confidence in the transformative potential of these therapies, driving innovation and paving the way for novel treatments across various diseases.
Facility Expansion for Cell and Gene Therapies Will Generate Surplus Worldwide T-Cell Therapy Market Opportunities
Expanding facilities for cell and gene therapies presents a remarkable opportunity in the biopharmaceutical and healthcare industries. With the rapid advancements in these transformative therapies, there is an escalating demand for their production and delivery to patients. Facility expansion not only enables meeting this growing demand but also offers several strategic advantages. Firstly, it allows for increased scalability, which is crucial as these therapies progress from clinical trials to commercialization. The ability to manufacture larger quantities of these therapies ensures broader patient access, addressing previously unmet medical needs. Moreover, facility expansion can lead to cost reductions through economies of scale, making these cutting-edge treatments more financially accessible for both patients and healthcare systems.
Secondly, expanding facilities is closely tied to regulatory compliance and quality assurance. Regulatory agencies, such as the FDA, require strict adherence to Good Manufacturing Practices (GMP) to ensure the safety and efficacy of cell and gene therapies. Expanding facilities involves investing in state-of-the-art manufacturing processes and quality control systems, which not only meets these regulatory requirements but also instills confidence in the therapies among healthcare providers, patients, and the broader medical community. This opportunity for facility expansion not only accelerates the commercialization of innovative treatments but also contributes to the overall growth and sustainability of the cell and gene therapy industry.
T-Cell Therapy Market Segmentation
The global market for T-cell therapy has been categorized into modality, therapy type, indication, end-user, and region
- The modaliy segment of the T-cell therapy market is split into research and commercialized
- Therapy type segment includes desktop, mobile, and headsets
- Technologies considered in the market are t-cell receptor (TCR)-based, CAR t-cell therapy, and tumor infiltrating lymphocytes (TIL)-based
- The indication segment is categorized into hematologic malignancies (leukemia, lymphoma, and myeloma), solid tumors (brain & central nervous system, melanoma, liver cancer, and others), and others
- Hospitals and cancer treatment centers are end-users profiled in the market
- The market is divided into five regions: North America, Asia-Pacific, Europe, Latin America, and the Middle East and Africa (MEA)
T-Cell Therapy Market Share
As per the T-cell therapy market forecast, the research modality sub-segment is expected to dominate the market share from 2023 to 2032.
In 2022, CAR t-cell therapy held a significant market share within the type segment, and this trend is expected to persist.
Hematologic malignancies are projected to lead the market in the coming years, while solid tumors are anticipated to gain significant traction from 2023 to 2032.
According to the T-cell therapy market analysis, hospitals end-user are the leading offering segment in 2022.
T-Cell Therapy Market Regional Outlook
The United States led the T-cell therapy market in North America. The FDA had approved several CAR-T cell therapies for blood cancers, making it a hub for research, development, and commercialization. The presence of major pharmaceutical companies and academic institutions in the region, combined with substantial investment in biotechnology, contributed to North America's dominant position in the T-cell therapy market.
The Asia-Pacific region, notably China and Japan, showed significant potential for T-cell therapy market growth. The region had a large patient population and an increasing focus on healthcare innovation. China, in particular, was becoming a major player in T-cell therapy research and development. The country had approved some CAR-T therapies, and Chinese companies were conducting clinical trials for various indications.
T-Cell Therapy Market Players
Some prominent T-cell therapy companies covered in the industry include Cartesian Therapeutics, Inc., Autolus Therapeutics, Immunocore Ltd., Celyad Oncology, Bristol-Myers Squibb Company, Caribou Biosciences, Inc, Novartis AG, Cellectis, Inc., Gilead Sciences, Inc., and Johnson & Johnson.
Click here to buy the Premium Market Research report https://www.acumenresearchandconsulting.com/buy-now/0/2494
Receive our personalized services and customization by clicking here https://www.acumenresearchandconsulting.com/request-customization/2494
Mr. Frank Wilson
Acumen Research and Consulting
USA: +13474743864
India: +918983225533