May 2024
The Global Chronic Obstructive Pulmonary Disease Treatment Market Size accounted for USD 19.7 Billion in 2023 and is estimated to achieve a market size of USD 31.9 Billion by 2032 growing at a CAGR of 5.6% from 2024 to 2032.
The Global Chronic Obstructive Pulmonary Disease Treatment Market Size accounted for USD 19.7 Billion in 2023 and is estimated to achieve a market size of USD 31.9 Billion by 2032 growing at a CAGR of 5.6% from 2024 to 2032.
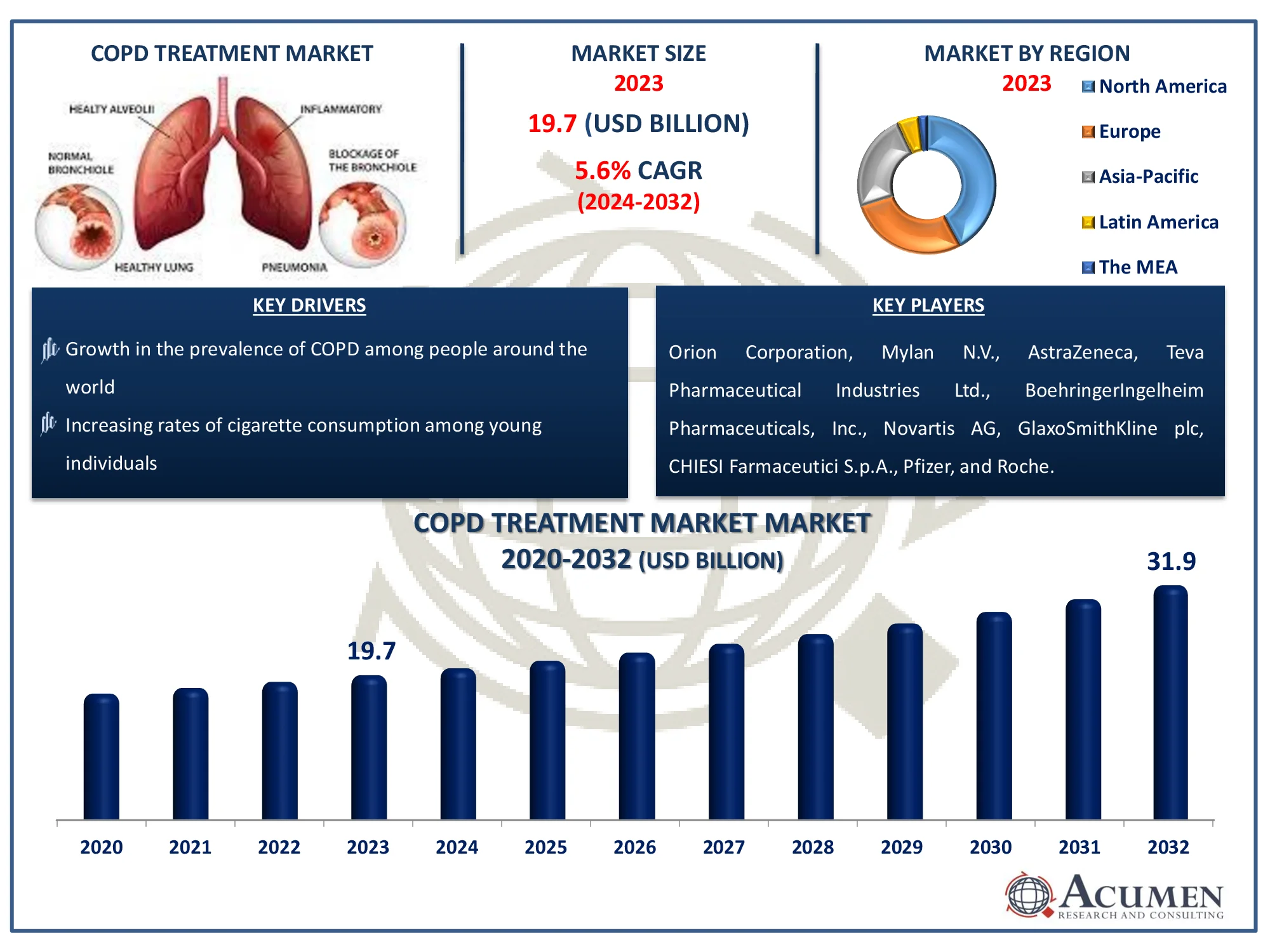
The rise in the prevalence of chronic obstructive pulmonary disease (COPD) is predicted to drive the global chronic obstructive pulmonary disease (COPD) treatment market growth. Furthermore, continuous R&D and product lines for the diagnosis of chronic obstructive pulmonary disease are likely to fuel the worldwide chronic obstructive pulmonary disease treatment market value in the coming years. Chronic obstructive pulmonary disease (COPD) is a kind of chronic inflammatory respiratory problem that impairs circulation from the lungs. COPD symptoms include issues with breathing, mucous production, coughing, and wheezing.
|
Market |
Chronic Obstructive Pulmonary Disease Treatment Market |
|
Chronic Obstructive Pulmonary Disease Treatment Market Size 2023 |
USD 19.7 Billion |
|
Chronic Obstructive Pulmonary Disease Treatment Market Forecast 2032 |
USD 31.9 Billion |
|
Chronic Obstructive Pulmonary Disease Treatment Market CAGR During 2024 - 2032 |
5.6% |
|
Chronic Obstructive Pulmonary Disease Treatment Market Analysis Period |
2020 - 2032 |
|
Chronic Obstructive Pulmonary Disease Treatment Market Base Year |
2023 |
|
Chronic Obstructive Pulmonary Disease Treatment Market Forecast Data |
2024 - 2032 |
|
Segments Covered |
By Type, By Drug Class, By Distribution Channel, and By Geography |
|
Regional Scope |
North America, Europe, Asia Pacific, Latin America, and Middle East & Africa |
|
Key Companies Profiled |
Orion Corporation, Mylan N.V., AstraZeneca, Teva Pharmaceutical Industries Ltd., BoehringerIngelheim Pharmaceuticals, Inc., Novartis AG, GlaxoSmithKline plc, CHIESI Farmaceutici S.p.A., Pfizer, and Roche. |
|
Report Coverage |
Market Trends, Drivers, Restraints, Competitive Analysis, Player Profiling, Covid-19 Analysis, Regulation Analysis |
The COPD treatment market value is driven by an increase in the numeral of COPD cases, an increase in FDA approval, and the continuous launch of a few products in the U.S. In addition, an increment in the geriatric populace and a rise in the use of pharmaceuticals in rising nations are alternate components adding to the development of the market. Nonetheless, the patent expiry of marked products and accessibility of conventional counterparts and elective treatment alternatives are foreseen to control the worldwide market.
High occurrence of COPD, promising medications in the pipeline, development of the pharmaceutical business in developing markets, for example, Asia-Pacific, and launch of products, for example, inhalers are anticipated to increase the worldwide COPD treatment market growth from the year 2024 to 2032. For instance according to the WHO Low- and middle-income nations account for about 90% of COPD fatalities among people under the age of 70.
An increase in the commonness of COPD, high environmental contamination, increment in the geriatric populace, and development of the pharmaceutical business are foreseen to fuel the development of the chronic obstructive pulmonary disease treatment market revenue in the Asia-Pacific. An article published in the International Journal of Pulmonary and Respiratory Sciences expressed that COPD is the third driving reason for death in India.
 Chronic Obstructive Pulmonary Disease Treatment Market Segmentation
Chronic Obstructive Pulmonary Disease Treatment Market SegmentationThe worldwide market for chronic obstructive pulmonary disease treatment is split based on type, drug class, distribution channel, and geography.
In 2023, the chronic bronchitis segment dominated the chronic obstructive pulmonary disease treatment market due to its higher prevalence among COPD patients. Persistent bronchitis is defined as persistent inflammation of the bronchial airways, which results in increased mucus production, coughing, and breathing difficulties. Smoking, air pollution, and occupational exposure to irritants are all common causes of the illness, which contributes to its prevalence. Bronchodilators, corticosteroids, and combination therapy are commonly used to relieve symptoms and improve lung function, which increases demand for related treatments. Furthermore, the increasing adoption of sophisticated drug delivery methods, as well as the availability of effective chronic bronchitis maintenance medicines, help to strengthen its market leadership. This tendency is expected to continue as awareness and availability to therapies increase.
According to a chronic obstructive pulmonary disease treatment industry analysis, the combination segment is expected to grow significantly in the market over the forecasting years. The combination segment has been segmented into long-acting beta-agonists & inhaled corticosteroids (LABA-ICS), triple therapy, long-acting muscarinic antagonist & inhaled corticosteroids (LAMA-ICS), and others. The bronchodilators segment has been bifurcated into short-acting beta agonist (SABA), long-acting beta-agonist (LABA), and long-acting muscarinic rival (LAMA). The combination segment is anticipated to represent the major share of the market by 2030 because of the increment in the number of doctors recommending combination treatment and the increase in the number of medications accessible for COPD treatment. The bronchodilators segment is foreseen to develop at a steady pace attributable to an increment in the appropriation of long-acting muscarinic rival (LAMA) drugs.
Based on the chronic obstructive pulmonary disease treatment market forecast, the hospital pharmacies segment is predicted to grow at the fastest rate throughout the forecast period. This is due to an increase in the number of patients with chronic respiratory diseases as well as lifestyle disorders, as well as the heightened awareness of these illnesses. Furthermore, the availability of different diagnostic and treatment centers, increased purchasing power, as well as the availability of highly skilled healthcare professionals are driving growth in the segment.
North America
Europe
Asia-Pacific
Latin America
The Middle East & Africa
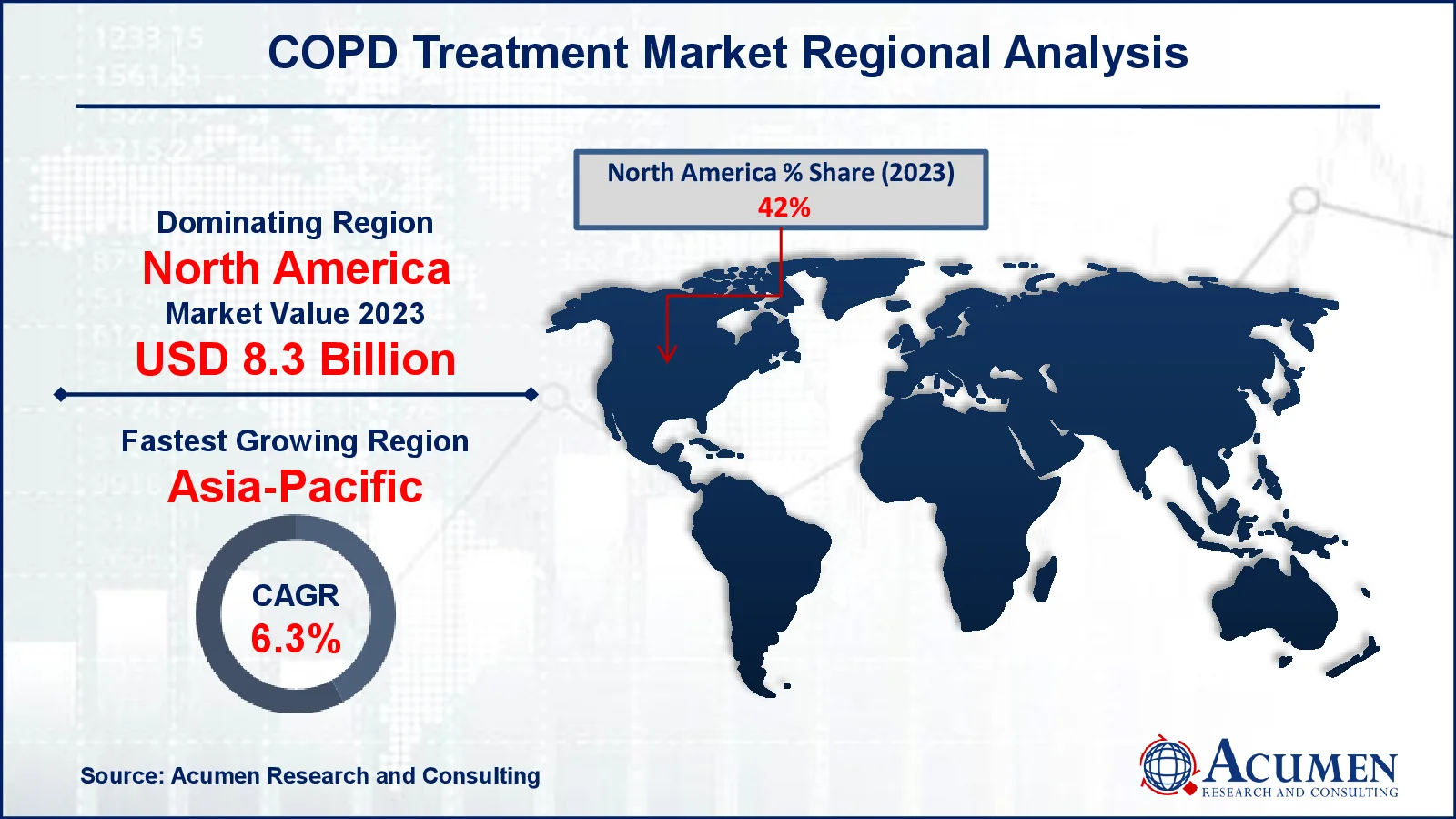 Chronic Obstructive Pulmonary Disease Treatment Market Regional Analysis
Chronic Obstructive Pulmonary Disease Treatment Market Regional AnalysisGeographically, North America is dominating the chronic obstructive pulmonary disease (COPD) treatment market due to a combination of factors, including high healthcare expenditure, advanced healthcare infrastructure, and a large patient population. The United States, in particular, has a high prevalence of COPD, driven by factors such as an aging population, smoking, and exposure to air pollution. This has led to significant demand for COPD treatments, driving the expansion of the regional chronic obstructive pulmonary disease treatment market share.
Furthermore, the availability of advanced healthcare facilities and the presence of leading pharmaceutical companies in North America have contributed to the dominance of the region in the COPD treatment market. North America is home to some of the world's largest pharmaceutical companies, which have invested heavily in the development of innovative COPD treatments. The region also has a well-established healthcare system, with advanced diagnostic and treatment options available to patients. This has resulted in better patient outcomes and improved quality of life for those living with COPD in North America.
Some of the top chronic obstructive pulmonary disease treatment companies offered in our report includes Orion Corporation, Mylan N.V., AstraZeneca, Teva Pharmaceutical Industries Ltd., BoehringerIngelheim Pharmaceuticals, Inc., Novartis AG, GlaxoSmithKline plc, CHIESI Farmaceutici S.p.A., Pfizer, and Roche.
Looking for discounts, bulk pricing, or custom solutions? Contact us today at [email protected]
May 2024
January 2025
May 2023
January 2020
Trusted Market Intelligence | 2026–2035 Insights | Designed to Support Faster, Smarter Decisions
Thank you! Our team will reach out to you soon.