January 2020
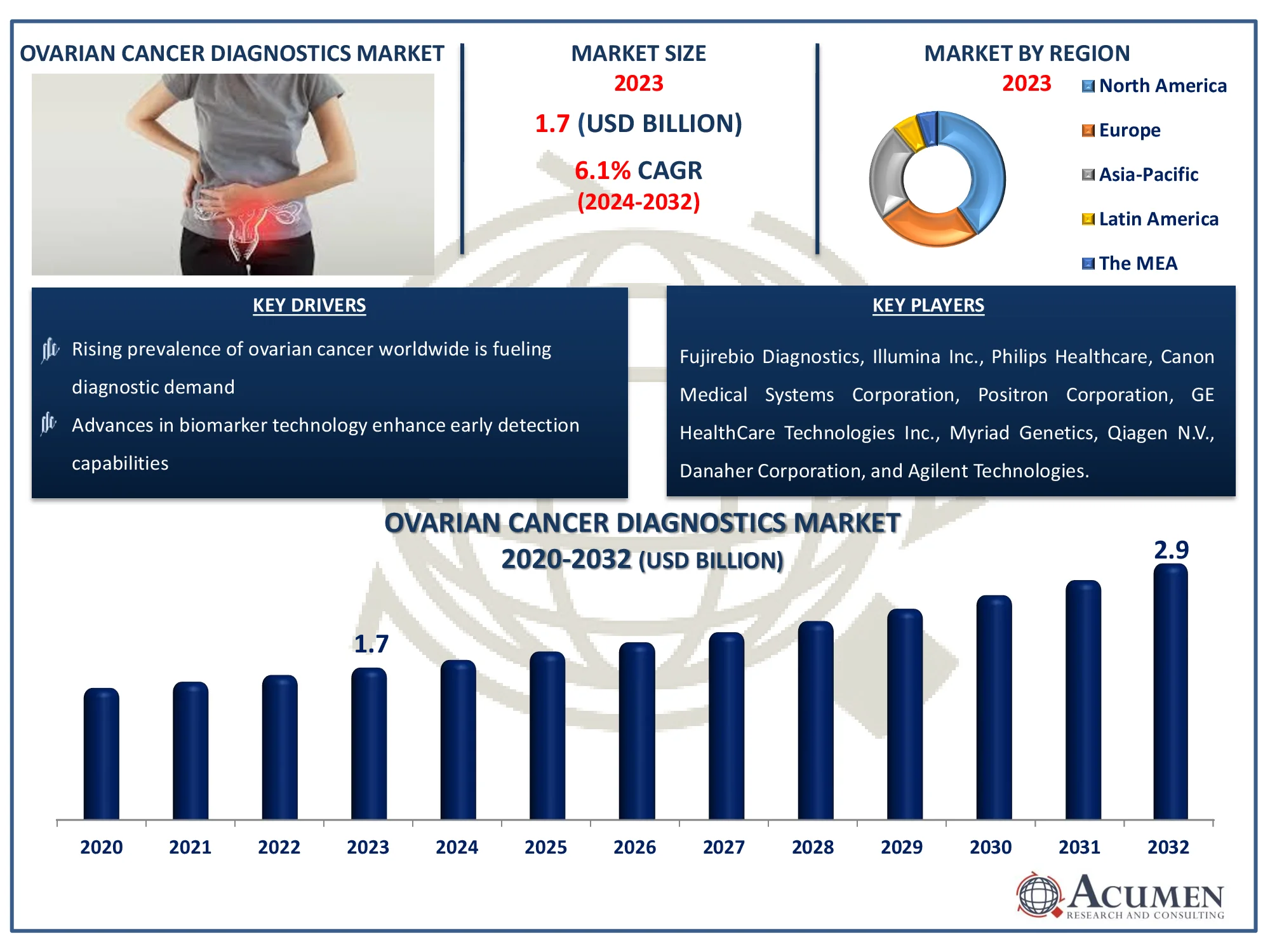
The Ovarian Cancer Diagnostics Market is expected to reach USD 2.9 billion by 2032, with a CAGR of 6.1%. Stay updated on market trends and growth factors.
The Global Ovarian Cancer Diagnostics Market Size accounted for USD 1.7 Billion in 2023 and is estimated to achieve a market size of USD 2.9 Billion by 2032 growing at a CAGR of 6.1% from 2024 to 2032.
Ovarian Cancer Diagnostics Market Highlights
One of the most prevalent kinds of cancer in females is ovarian cancer. It is a cancer which begins in the ovaries. Ovary is a woman reproductive organ that contains eggs or ova. There are two ovaries in the woman reproductive scheme: one on either side of the uterus. Hormones include progesterone and estrogen is produced by ovaries. Eggs are also produced by ovaries. Ovarian cancer can only be detected when it reaches the abdomen and pelvis. The ovarian cancer is more deadly and difficult to cure at this point. In the initial stage, it can be treated more effectively if it is limited to the ovarian cells.
Global Ovarian Cancer Diagnostics Market Dynamics
Market Drivers
Market Restraints
Market Opportunities
Ovarian Cancer Diagnostics Market Report Coverage
| Market | Ovarian Cancer Diagnostics Market |
| Ovarian Cancer Diagnostics Market Size 2022 |
USD 1.7 Billion |
| Ovarian Cancer Diagnostics Market Forecast 2032 | USD 2.9 Billion |
| Ovarian Cancer Diagnostics Market CAGR During 2023 - 2032 | 6.1% |
| Ovarian Cancer Diagnostics Market Analysis Period | 2020 - 2032 |
| Ovarian Cancer Diagnostics Market Base Year |
2022 |
| Ovarian Cancer Diagnostics Market Forecast Data | 2023 - 2032 |
| Segments Covered | By Diagnosis Type, By Cancer Type, By End-Use, And By Geography |
| Regional Scope | North America, Europe, Asia Pacific, Latin America, and Middle East & Africa |
| Key Companies Profiled | Fujirebio Diagnostics, Illumina Inc., Philips Healthcare, Canon Medical Systems Corporation, Positron Corporation, GE HealthCare Technologies Inc., Myriad Genetics, Qiagen N.V., Danaher Corporation, Agilent Technologies, F. Hoffmann-La Roche Ltd., and Abbott Laboratories. |
| Report Coverage |
Market Trends, Drivers, Restraints, Competitive Analysis, Player Profiling, Covid-19 Analysis, Regulation Analysis |
Ovarian Cancer Diagnostics Market Insights
The growing incidence and emphasis on early diagnosis & therapy of ovarian disease should boost business development. The illness has generic signs, including bloating, menstruation abnormality, vaginal swelling, indigestion and tiredness. No specific symptoms make it increasingly difficult to diagnose the disease early. The prognosis of diseases continues low, since most patients are diagnosed in developed phases. So comprehensive study is essential to comprehend the biology of diseases so that efficient therapy choices are developed with the aim of overcoming opposition and minimizing toxicity. Further development of biomarkers could improve the identification of patients who require maintenance.
Factors such as an increase in government projects and financing for ovarian cancer research should fuel the industry. For example, the non-profit Ovarian Cancer Research Alliance (OCRA) focuses on the R&D of ovarian cancer. It promotes several study programs related to the diagnosis and therapy of this disease.
Some key factors restricting growth are lack of awareness of exact causative factors and late diagnosis of the disease. The fundamental molecular processes to recognize major genes or biochemical pathways that can be used for diagnosis & therapy are highly unmet. However, growing healthcare spending and technological advances create growth possibilities for the entire industry.
Ovarian Cancer Diagnostics Market Segmentation
The worldwide market for ovarian cancer diagnostics is split based on diagnosis type, cancer type, end-use, and geography.
Ovarian Cancer Diagnostics Market By Diagnosis Type
According to ovarian cancer diagnostics industry analysis, imaging methods accounted for the greatest share in 2023 due to the growing significance of early treatment and cancer staging. The preliminary imaging technique for internally viewing and measurement of a tumor remains the transvaginal ultrasound. CT scans are chosen for the stadium of cancer, but are restricted to tiny tumors. Advanced imagery techniques, like MRI and PET, are used to detect metastases and severity of diseases.
The blood test industry is predicted to see steady growth in technology and increased customer consciousness throughout the forecast period. Ovarian cancer can contribute to high concentrations of some biomarkers that can function as tumor markers for diagnosis. For example, for this illness,CA-125 is the most prevalent blood exam. A greater market penetration for HER2, BRCA and KRAS mutation studies led to increased focus on genetic consulting and testing.
Ovarian Cancer Diagnostics Market By Cancer Type
The sort of ovarian cancer depends on where cancer cells develop. In this section, epithel tumors stayed a precursor, representing more than 91% of all ovarian malignancies, in 2023. Serous, mucinous, endometrioid, or clear cell can be epithelial tumors. After the cancer has metastasized within the peritoneal nucleus, most individuals with epithelial cell carcinoma are identified. In the developed phase, 70% to 80% of people are diagnosed and the therapy choices and survival rate are reduced considerably. Collectively, germ and stromal cell tumors are capturing a lower share of this industry. Stromal cells grow in hormones such as estrogen, progesterone and testosterone in the organs accountable for generating them. Although common, germ cell tumors are most common in females between the ages of 20 and 30 years.
Ovarian Cancer Diagnostics Market By End-Use
The biggest share in 2023 was retained by end-use labs of hospitals. This sector development can be ascribed to increased patient inflow for cancer treatment in clinics, increased customer consciousness and increased infrastructure expenditure in emerging and underdeveloped nations. Due to increased financing for cancer studies globally, the market for study institutes is anticipated to develop at the largest pace during the prediction era. The Fred Hutchinson Cancer Research Center, for example, is devoted to biomarkers and ovarian cancer screening.
Ovarian Cancer Diagnostics Market Regional Outlook
North America
Europe
Asia-Pacific
Latin America
The Middle East & Africa
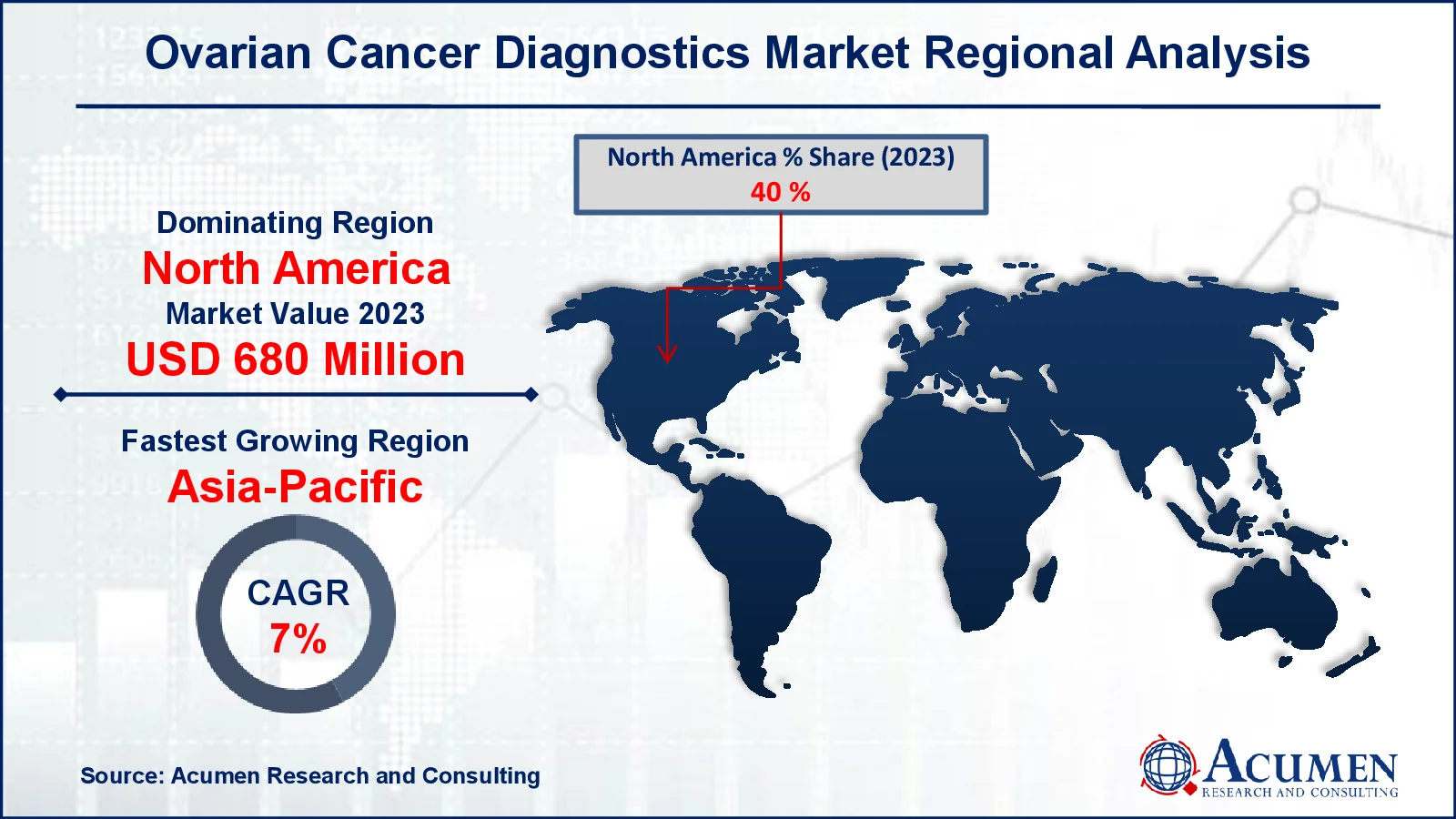
Ovarian Cancer Diagnostics Market Regional Analysis
North America accounted for over 40% of the total industry in 2023 due to the powerful business performance of US ovarian diagnostics. Some other variables contributing to ovarian cancer diagnostics market growth in this region are favorable public policies, increased healthcare spending and the existence of important producers. During the forecast period, Asia-Pacific is anticipated to develop at the most high level because of variables such as general economic development, improved health infrastructure, increased disposable income and increasing customer consciousness. Ovarian cancer diagnostics are anticipated to grow rapidly in emerging countries like China, India and Indonesia during the ovarian cancer diagnostics market forecast period. This is attributed to the significant rise of ovarina cancer patients in China (61,060), India (47,333), and Indonesia (15,130) as per the data reveled from the World Cancer Research Fund 2022.
Ovarian Cancer Diagnostics Market Players
Some of the top ovarian cancer diagnostics companies offered in our report includes Fujirebio Diagnostics, Illumina Inc., Philips Healthcare, Canon Medical Systems Corporation, Positron Corporation, GE HealthCare Technologies Inc., Myriad Genetics, Qiagen N.V., Danaher Corporation, Agilent Technologies, F. Hoffmann-La Roche Ltd., and Abbott Laboratories.
Looking for discounts, bulk pricing, or custom solutions? Contact us today at sales@acumenresearchandconsulting.com
January 2020
June 2022
January 2021
December 2022