June 2024
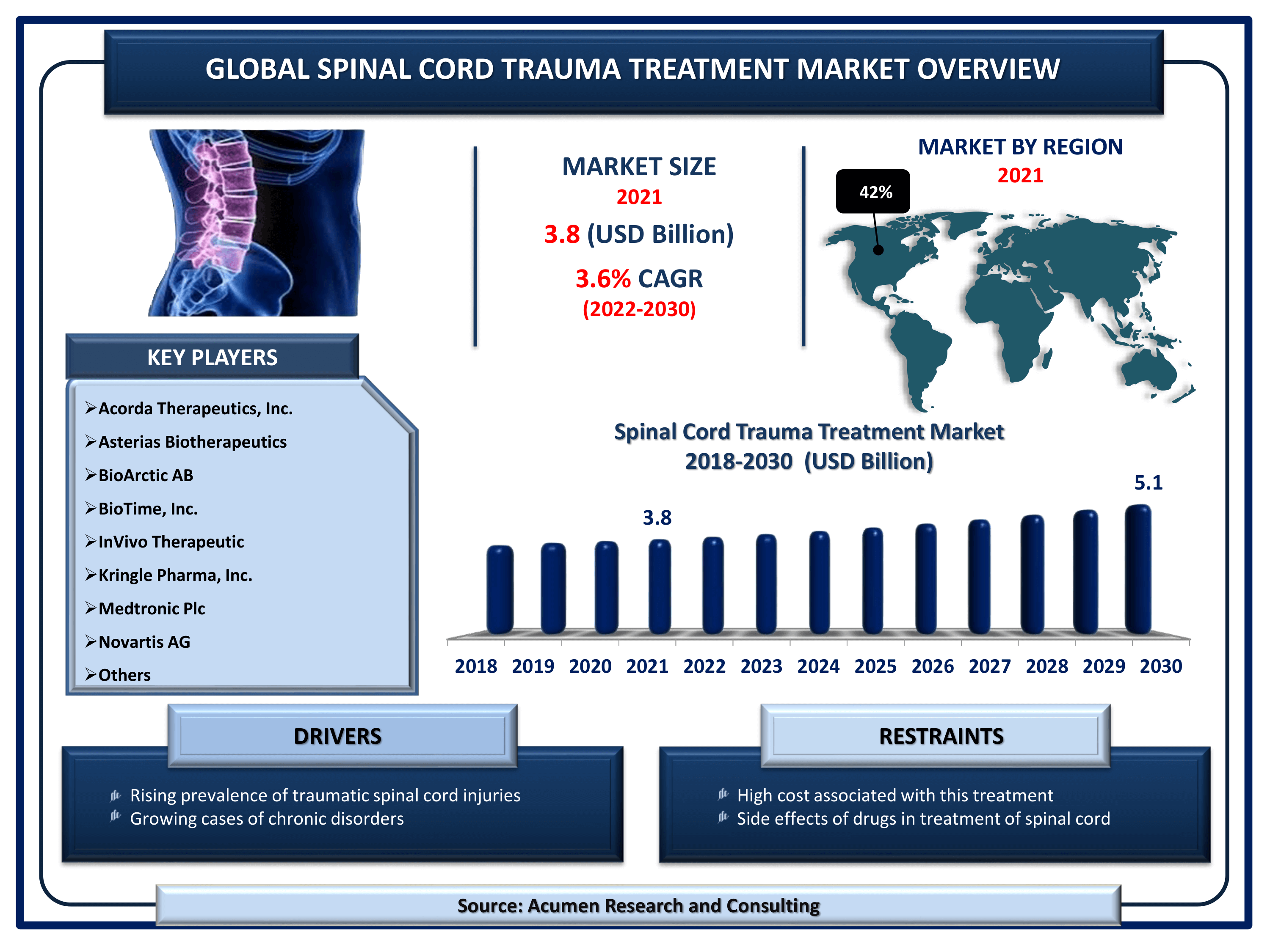
Spinal Cord Trauma Treatment Market Size accounted for USD 3.8 Billion in 2021 and is estimated to garner a market size of USD 5.1 Billion by 2030 rising at a CAGR of 3.6% from 2022 to 2030.
The Global Spinal Cord Trauma Treatment Market Size accounted for USD 3.8 Billion in 2021 and is estimated to garner a market size of USD 5.1 Billion by 2030 rising at a CAGR of 3.6% from 2022 to 2030. Growing incidences of spinal cord injuries throughout the world are fueling the spinal cord trauma treatment market growth. In addition to that, the rising elderly population increased base of chronic patients, and growth in the number of car accidents are also supporting the spinal cord trauma treatment market value. Furthermore, favorable reimbursement policies and growing support from the government are some leading spinal cord trauma treatment market trends that are reinforcing the industry growth in the coming years.
Spinal Cord Trauma Treatment Market Report Key Highlights
Damage to the spinal cord can occur in several ways; the most common cause being external trauma. A spinal cord injury is damage to any part of the spinal cord or nerves at the end of the spinal canal (cauda equina) that often causes permanent changes in strength, sensation, and other body functions below the site of injury. Spinal cord injury tends to affect more men as compared to women and the majority of the people who sustains a spinal cord injury are between the ages of 16 and 30 due to the increased likelihood of risky behavior. Currently, there are no ways to reverse damage to the spinal cord. However, researchers are continuously working on new treatments, involving prostheses and medications that may promote the regeneration of nerve cells or improve the function of the nerves that remain after a spinal cord injury (SCI). According to the World Health Organization (WHO), there is no real estimate of global prevalence, but estimated annual global incidences are 40 to 80 cases per million population. Up to 90% of these cases are caused due to traumatic cases though the population of non-traumatic spinal cord injury appears to be growing.
Global Spinal Cord Trauma Treatment Market Dynamics
Market Drivers
Market Restraints
Market Opportunities
Spinal Cord Trauma Treatment Market Report Coverage
| Market | Spinal Cord Trauma Treatment Market |
| Spinal Cord Trauma Treatment Market Size 2021 | USD 3.8 Billion |
| Spinal Cord Trauma Treatment Market Forecast 2030 | USD 5.1 Billion |
| Spinal Cord Trauma Treatment Market CAGR During 2022 - 2030 | 3.6% |
| Spinal Cord Trauma Treatment Market Analysis Period | 2018 - 2030 |
| Spinal Cord Trauma Treatment Market Base Year | 2021 |
| Spinal Cord Trauma Treatment Market Forecast Data | 2022 - 2030 |
| Segments Covered | By Treatment Type, By Injury, By End-User, And By Geography |
| Regional Scope | North America, Europe, Asia Pacific, Latin America, and Middle East & Africa |
| Key Companies Profiled | Acorda Therapeutics, Inc., Asterias Biotherapeutics, BioArctic AB, BioTime, Inc., InVivo Therapeutic, Kringle Pharma, Inc., Medtronic, Novartis AG, Pfizer Inc., Pharmicell Co. Ltd., and ReNetX Bio. |
| Report Coverage |
Market Trends, Drivers, Restraints, Competitive Analysis, Player Profiling, Regulation Analysis |
Spinal Cord Trauma Treatment Market Insights
Presence of the Stellar Industry Enhances the Growth of the Spinal Cord Trauma Treatment Market
Continual research and the presence of a stellar industry for global healthcare enhance the spinal cord injury treatment market growth. The stellar study is a phase 3 clinical study that is designed to evaluate the efficacy and safety of Eflornithine in combination with Lomustine compared to Lomustine taken alone in patients suffering from Anaplastic Astrocytoma i.e. recurrence or progression after radiation therapy and Temozolomide (Temodar®) chemotherapy. Anaplastic Astrocytoma originates from astrocytes, a type of cell that normally wraps and protects nerves in the brain and spinal cord. Unfortunately, the exact cause of Anaplastic Astrocytoma is not known. On the other hand, Eflornithine acts as a molecule that targets a key enzyme called ornithine decarboxylase. According to the studies conducted by the American Brain Tumor Association on humans and animals, Eflornithine has shown a good response towards delaying or stoppage the growth of brain cancer as well as other kinds of cancer cells.
Cell Pores Discovery: A New Hope for the Patients with Spinal Cord Injury
Scientists have discovered a new treatment that dramatically reduces swelling after brain and spinal cord injuries. The new treatment is developed by a team of international scientists working at Aston University and the University of Birmingham, (UK), Harvard Medical School (US), University of Calgary (Canada), Lund University (Sweden), and Copenhagen University (Denmark). The researchers used existing licenses of anti-psychotic medicine trifluoperazine (TFP). Testing the treatment on injured rats, it was found that those animals that are given a single dose of the drug at the trauma site recovered full movement and sensitivity in less than two weeks. The treatment works by counteracting the cells normal reaction to a loss of oxygen caused by trauma in the CNS, the brain, and the spinal cord. TFP is already licensed for use in humans by the US Federal Drug Administration (FDA) and the UK National Institute for Health and Care Excellence (NICE).
Rising Incidences of Sports Injuries Bolster the Growth of the Global Spinal Cord Trauma Treatment Market
According to the Australian Institute of Health and Welfare, the 3 most often associated sports-related spinal injuries are water sports, wheeled motor sports, and equestrian activities. The average acute-care cost of the first admission record for a cervical SCI was US$ 67,523 which was the highest for any type of sports-related injury. A thoracic lumbosacral SCI was the second most expensive type of sports-related injury at an average cost of US$ 37,797 per person. Just one-quarter (26%) of the traumatic SCI cases reported to the ASCIR for 2015-2016, occurred while the person engaged in sports or leisure activities is highly prevalent in males than women which is 89% (58 of 65 cases). Moreover, 20% of the SCI were reported due to water-related event whereas pedal cyclists contribute the greatest proportion to sport and leisure SCI cases (14 cases or 22%) while motorcyclist contributes 15%.
Spinal Cord Trauma Treatment Market Segmentation
The worldwide spinal cord trauma treatment market is split based on treatment type, injury, end-user, and geography.
Spinal Cord Trauma Treatment Market By Treatment Type
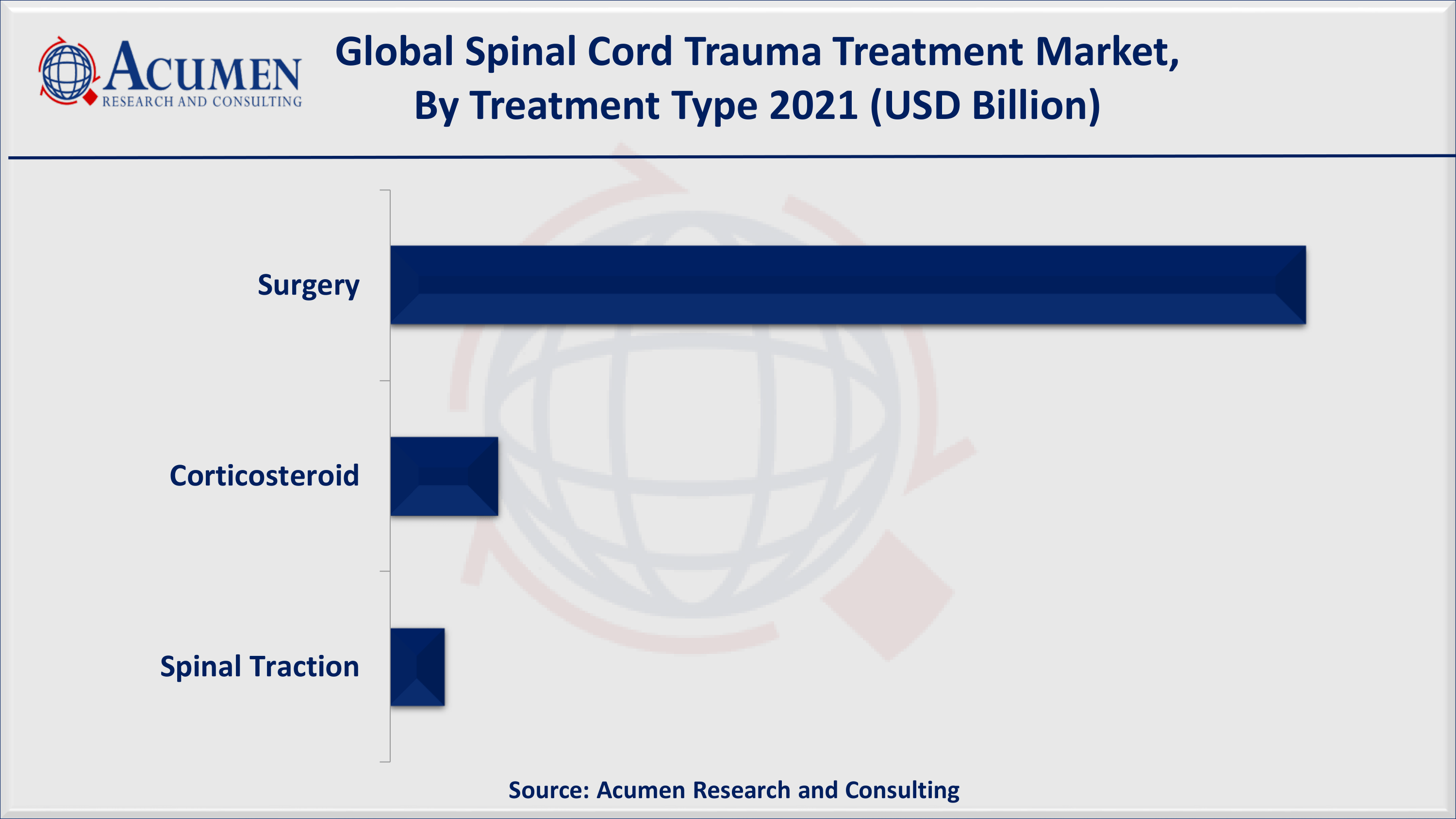
Surgery accounted for a significant market revenue in 2021, according to our spinal cord trauma treatment industry analysis. Howeer, corticosteroid is likely to gain significant traction in the coming years. After the initial tissue damage associated with spinal cord injury, the inflammatory response is controllable. The inflammatory response is conducted by tissue ischemia, oxidative stress, and immune response. Steroids such as methylprednisolone are utilized in the reduction of oxidative stress and act as a primary degenerative event post-trauma. Every year, about 40 million people suffer globally from spinal cord injury as per the estimates released by Cochrane. Several drugs are prescribed to patients that reduce the extent of permanent paralysis. All the research conducted in all seven trials involves just one steroid effective that is methylprednisolone. The results show that the treatment with the steroid methylprednisolone does improve movement but it should start soon after the injury has happened within no more than eight hours. It should be continued for 24 to 48 hours. The treatment offers the patient a normal amount of movement and more research is necessary on steroids in combination with other drugs.
Spinal Cord Trauma Treatment Market By Injury
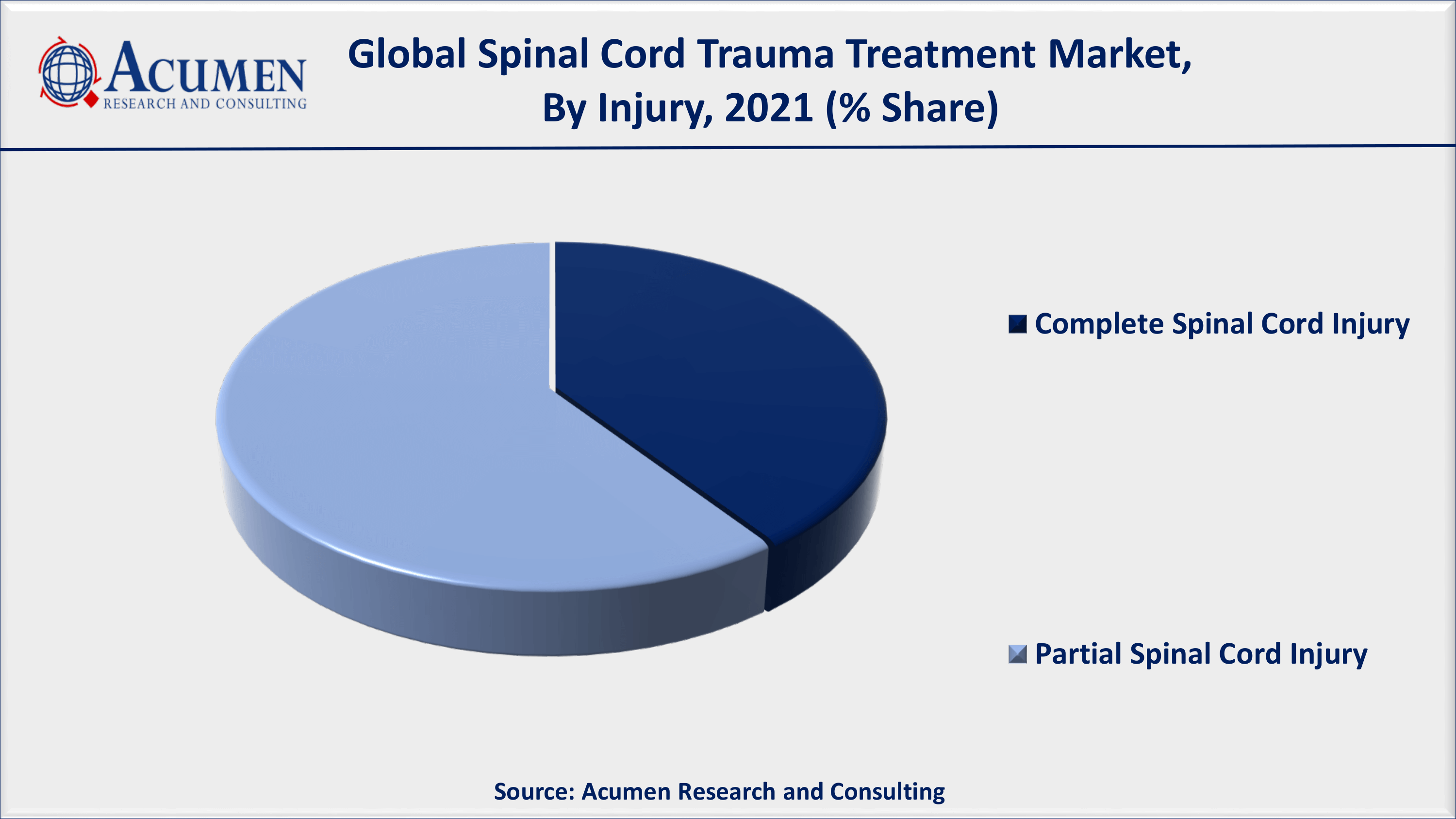
Complete spinal cord injury accounted for a sizable market share in 2021 and is expected to continue to do so in the coming years. The increasing number of severe spinal cord injuries caused by road accidents, gunshot wounds, as well as sport injuries can cause to complete spinal cord injuries. As a result, this sub-segment will lead the market till 2030. However, the partial spinal cord injuries is likely to attain a significant growth rate in the coming years.
Spinal Cord Trauma Treatment Market By End-User
According to our spinal cord trauma treatment market forecast, the trauma centers end-user subsegment accounted for a noteworthy market share from 2022 to 2030. The high growth in the segment can be attributed to the presence of emergency treatments for trauma patients. In addition to that, hospitals accounted for an impressive spinal cord trauma treatment market share and growth rate from 2022 to 2030.
Spinal Cord Trauma Treatment Market Regional Outlook
North America
Europe
Latin America
Asia-Pacific
The Middle East & Africa (MEA)
North America recorded largest market share followed by Europe in Spinal Cord Trauma Treatment Market
Availability of treatment options in North America for spinal cord trauma treatment enhances the regional market growth. Corticosteroid and spinal traction have gained significant attention in North America region owing to rising incidences of spinal cord injuries (SCI). Additionally, the North America regional market is projected to expand as people from across the world travel to the US for better treatments. According to the National Institute of Neurological Disorders and Stroke, it is estimated that 12,000 spinal cord injuries are recorded annually in the US alone. Moreover, more than a quarter of a million Americans are currently living with spinal cord injuries the cost of managing the care of spinal cord injury patient is US$ 3 Bn per year. Tech advancements for diagnosis and rehabilitation centers are well established in the US have gained huge prominence globally for people travelling for the treatment of spinal cord traumatic cases.
Spinal Cord Trauma Treatment Market Players
The global spinal cord trauma treatment companies profiled in the report include Acorda Therapeutics, Inc., Asterias Biotherapeutics, BioArctic AB, BioTime, Inc., InVivo Therapeutic, Kringle Pharma, Inc., Medtronic, Novartis AG, Pfizer Inc., Pharmicell Co. Ltd., and ReNetX Bio.
Looking for discounts, bulk pricing, or custom solutions? Contact us today at [email protected]
June 2024
August 2024
July 2024
February 2020
Trusted Market Intelligence | 2026–2035 Insights | Designed to Support Faster, Smarter Decisions
Thank you! Our team will reach out to you soon.